Chemicals and Services
Cooling Tower Water Analysis
Water is an important raw material in cooing tower industry due to the following properties:
- Abundant
- Readily available
- Cheap
- Easy to handle
- high heat capacity
- incompressible at normal conditions
- does not decompose
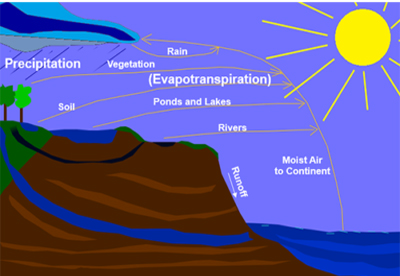
Although, the disadvantage in using water is that it dissolves almost everything that it touches, including metals and stones.The process in which rain water falls to earth is called the hydrologic cycle. Water from rain becomes ground water, and enters bodies of water like ponds, lakes, rivers and oceans. This then evaporates back to the atmosphere and becomes rain again.
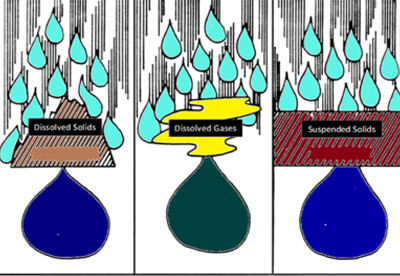
- Rain dissolves all the minerals in three different forms:
- Gases (oxygen and carbon dioxide)
- Dissolved solids (dissolved minerals)
- Suspended solids (clay,silt, dirt and oil)
- There are two sources of water for industrial plant use. These waters are very different in the impurities they contain. Both water’s dissolving ability and the dissolved solids themselves are harmful to the cooling system equipment. Unless properly treated, water will cause scale, corrosion, and fouling.
- Surface water
- low dissolved solids
- high suspended solids
- abrupt changes in quality with seasons and weather changes
- Ground water
- high dissolved solids
- low suspended solids
- high iron and manganese content
- low oxygen content, may contain sulfide gas
- relatively constant quality and temperature
Liang Chi Chemicals and Technical Services Division offers water analysis for different samples including the following:
- make-up water for cooling tower
- recirculating water for cooling tower
- chilled water
- boiler water
- templifier water
The important parameters that need to be tested include:
- It is the measure of how well water will “conduct” an electrical current. It is important to measure conductivity as a reference for the cycles of concentration (COC) of the system.
- It comprises calcium and magnesium that make water hard to use in washing.
- Total Alkalinity. It is the measure of water’s buffering capacity. It comprises bicarbonates (HCO3–) carbonates (CO32-) and hydroxide (OH–). If alkalinity is too high, scaling deposits may form. If the alkalinity is too low, corrosion may form.
- OH– Alkalinity. It is the measure of alkalinity in the form of hydroxide ions. High value may result to brittleness of boiler tubes.
- pH. It measures the amount of hydrogen ion present in water. A pH greater than 7 is considered acidic and a pH less than 7 is considered to be basic.
- Iron. It is the measure of dissolved Fe2+ It is important to analyse this parameter to determine if there is iron deposition along the system.
- Copper. It is also important to measure copper ions to determine the deposition along the system, which may be attributed to acidic property of water.
- Silica. It is commonly found in various living organisms, which may enter the cooling tower system through make-up water.
- Residual phosphonate. The residual phosphonate must be maintained in order to prevent scaling and fouling for open type cooling systems.
- Residual nitrite. The residual nitrite must be maintained in order to prevent scaling and fouling for closed type cooling systems.
MICROBIOLOGICAL ANALYSIS
- Legionella pneumophila. This causes Legionnaires’ disease and may be present in surface waters and adjacent soils.
Langelier Saturation Index (LSI) is the key to determining the scaling or corrosion tendency of water. The three factors needed to compute LSI are: Calcium hardness, M-alkalinity and pH. The higher the pH, the greater the potential is for scale formation. The lower the pH, the greater the potential is for corrosion.
LCIPI also provides on-site analysis. It houses two laboratories in Taytay and Cebu sites. Its laboratory analysts provide excellent analysis to regularly check the water conditions. Its chemical engineers constantly monitor the parameters and provide timely recommendations for problems encountered by cooling tower clients.


